Utilizing CRISPR-Cas13d-knockdown in zebrafish to study a rare monogenic bone fragility syndrome
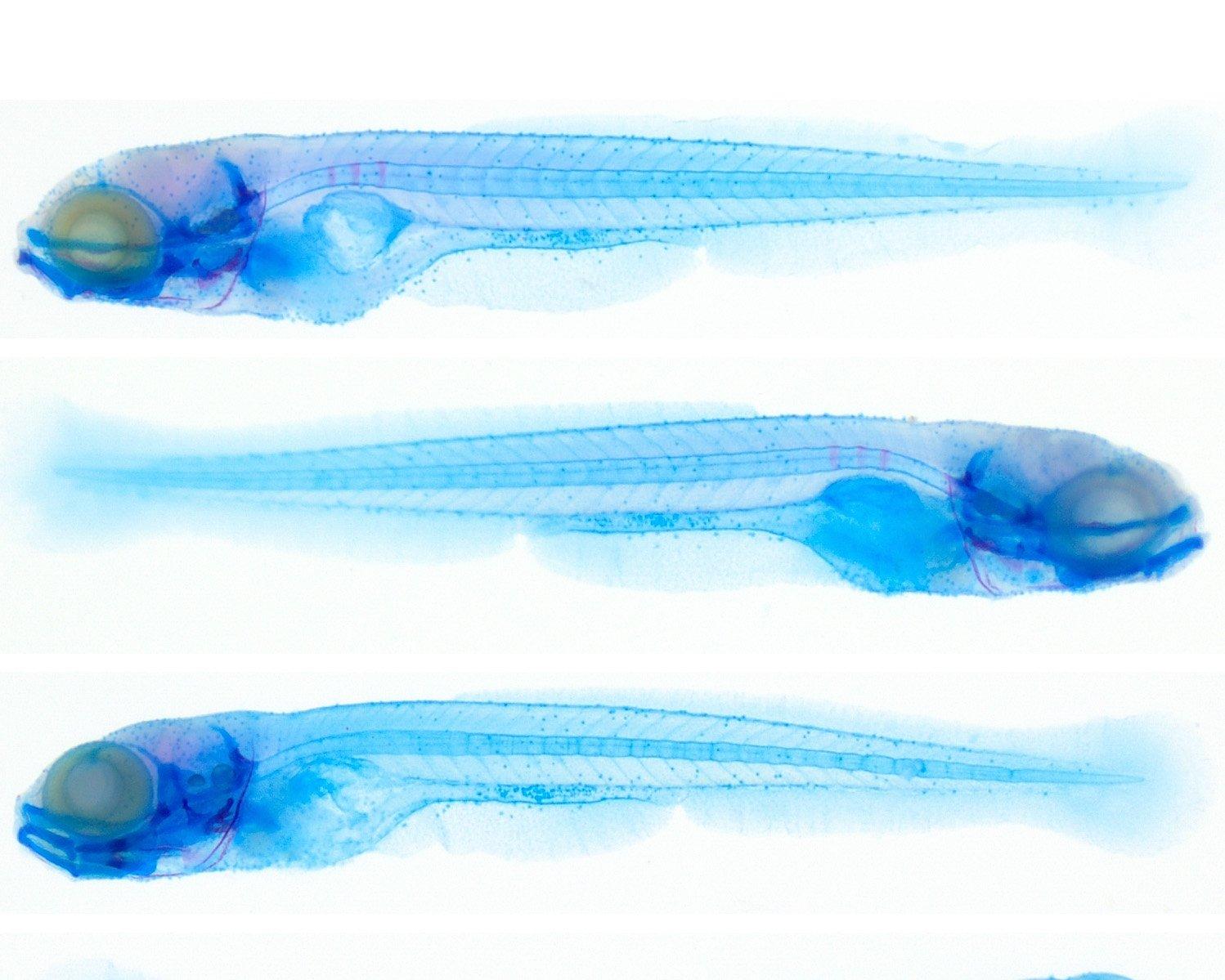
Osteoporosis is a multifactorial disease with a strong genetic component. Pathogenic variants in the SGMS2 gene cause a rare form of primary osteoporosis, “Calvarial doughnut lesions with bone fragility” (CDL). Since the mechanisms by which disrupted SGMS2 lead to skeletal fragility are still largely unknown, we set out to explore the gene’s function in a zebrafish model. Zebrafish has two orthologs of the human SGMS2 gene, sgms2a and sgms2b.
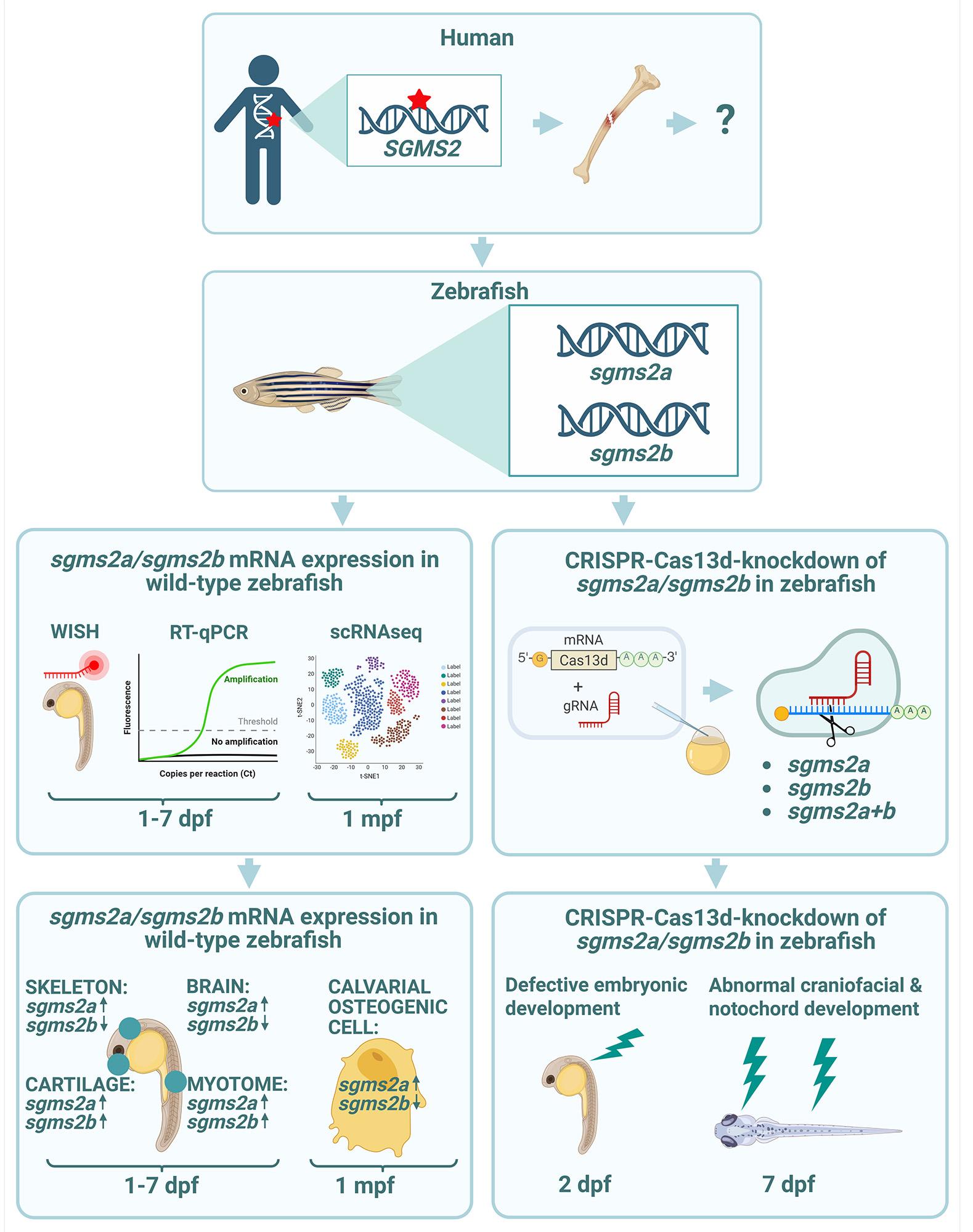
First, we studied expression profiles of both sgms2a and sgms2b genes in a wild-type zebrafish at different developmental stages. Second, we made both genes non-functional during the early days of development and analyzed the outcome. We discovered that both genes are expressed in muscle and skeletal tissue during early development and sgms2a gene is only expressed in calvarial bone cells during the juvenile stage. We also observed that when sgms2 genes are non-functional, zebrafish embryos develop defectively and present with deformed notochord and craniofacial structures.
Our findings from zebrafish studies provide new information on the SGMS2 gene’s function and its role in early musculoskeletal development. The obtained zebrafish model may prove helpful in understanding the disease mechanisms in SGMS2-related osteoporosis (CDL) and in discovering new therapies for osteoporosis.
Original article:
Utilizing CRISPR-Cas13d-knockdown in zebrafish to study a rare monogenic bone fragility syndrome.
Määttä K, Chen YC, Pihlström S, Mäkitie RE, Dambroise E, Legeai-Mallet L, Panula P, Mäkitie O, Pekkinen M. Journal of Bone and Mineral Research. 2025.
Highlights 2025

Parenting style and practices can shape children's vegetable consumption
Children ate less vegetables when parents were more overprotective.

Myosin inhibition partially rescues the myofibre proteome in X-linked myotubular myopathy

Frailty outperforms conventional risk factors for predicting complications and death in type 1 diabetes

Cognitive performance in late midlife as a predictor of frailty over 17 years: A longitudinal birth cohort study

Sense of community and social participation underlying active aging among senior housing residents
As people live longer, many move into senior housing communities where they can receive support while maintaining independence. But what actually helps older adults stay active and engaged in everyday life? This study explored how a sense of community and social participation are linked to active aging among residents in senior housing.