Liver‑directed gene therapy shows strong therapeutic potential in mouse model of GRACILE syndrome
A new study from the Kallijärvi group demonstrates that targeting the liver alone with viral gene therapy prevents metabolic collapse, stabilizes blood glucose, improves growth and nearly doubles the survival in a mouse model of mitochondrial complex III deficiency. The work, led by Rishi Banerjee and colleagues, represents a major step forward for gene therapy approaches in primary mitochondrial diseases.
Although the understanding of mitochondrial disease mechanisms has advanced rapidly, progress toward effective treatments has been slow. For inherited genetic diseases, correcting or compensating for the underlying mutation - known as gene therapy or gene replacement - offers one of the most promising routes to meaningful clinical benefit. However, pre-clinical development is challenging as mitochondrial diseases often affect multiple organs, and physiologically relevant animal models are scarce. To date, only one primary mitochondrial disease has reached a gene‑replacement clinical trial.
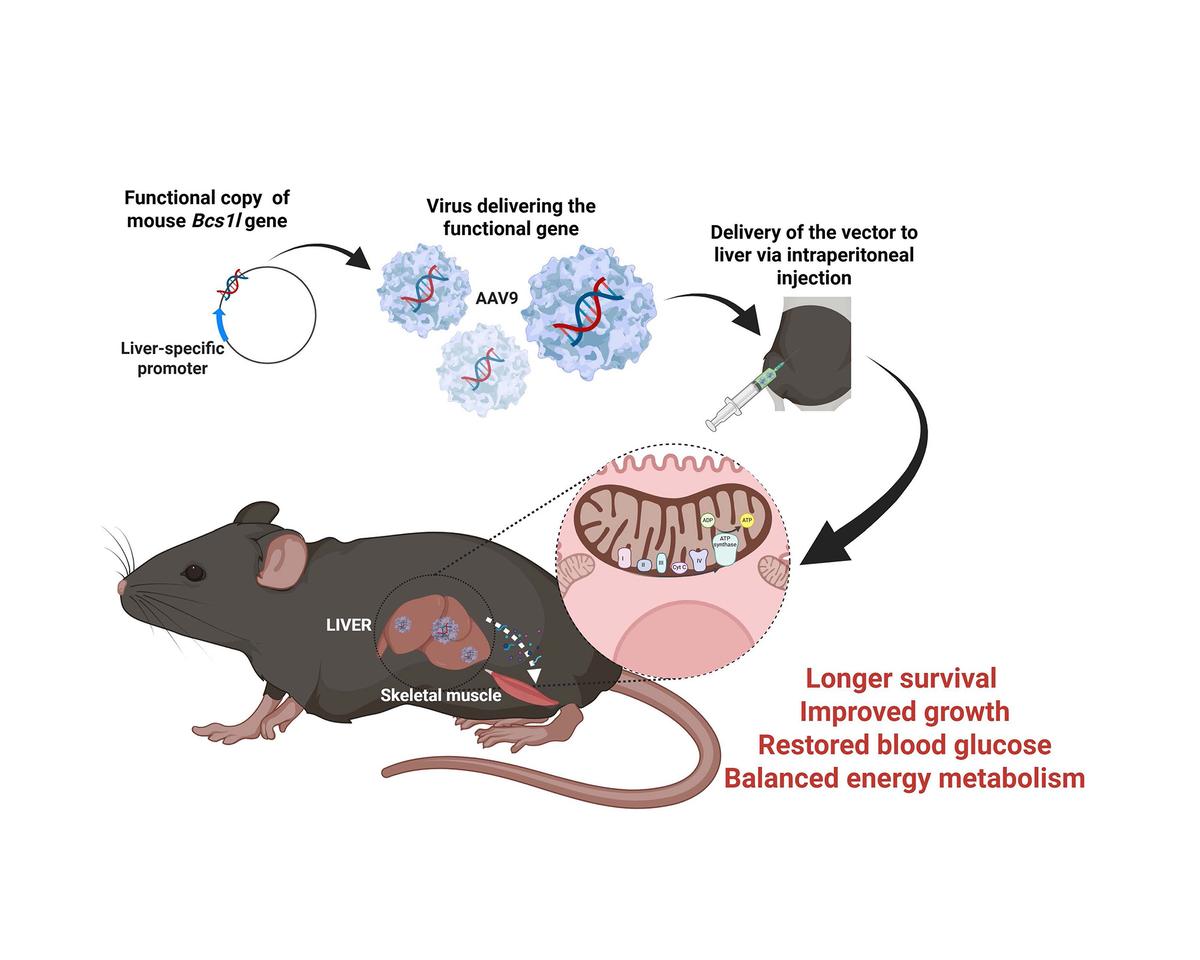
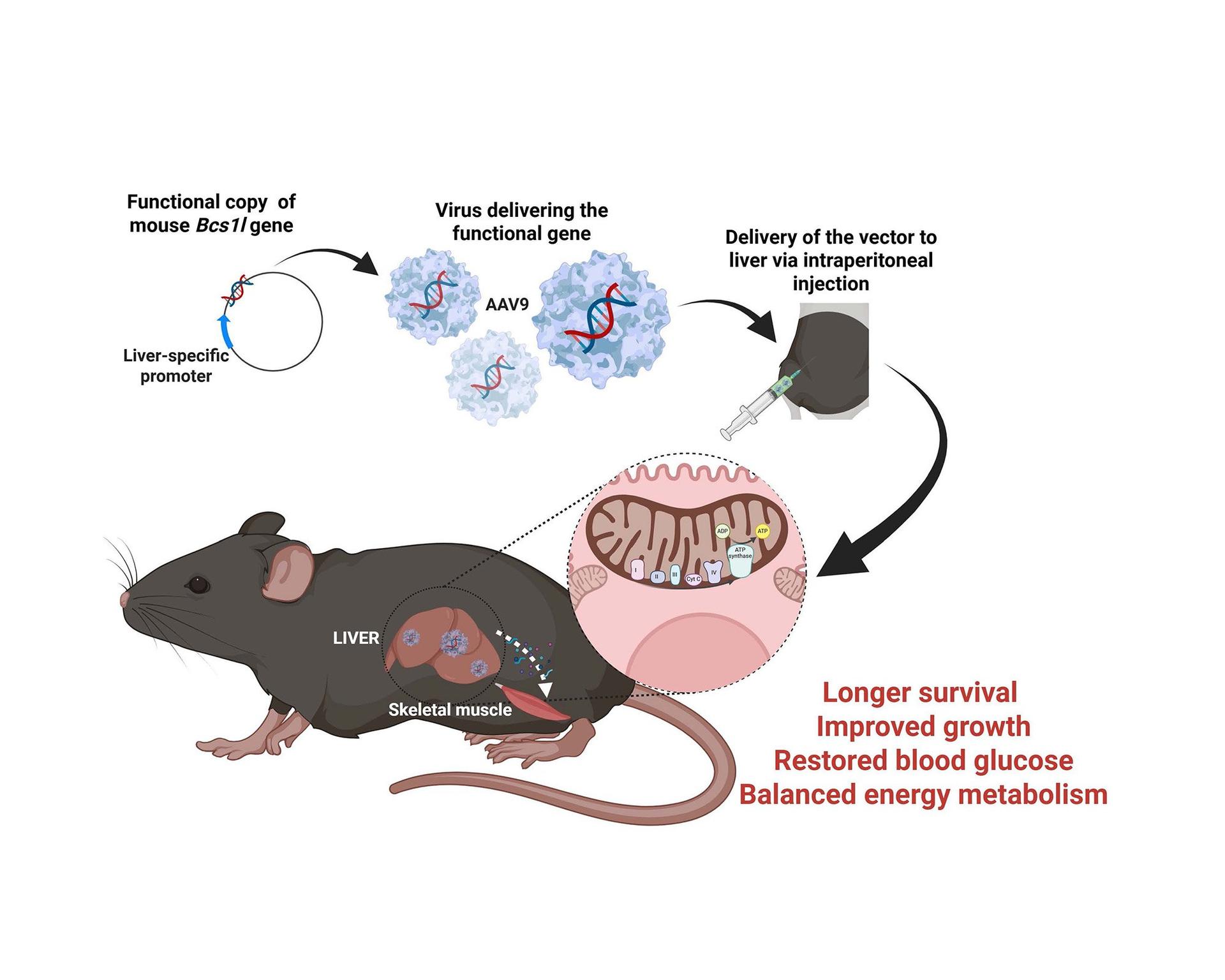
Using the group’s well‑established GRACILE syndrome mouse model (Bcs1l-p.S78G knock-in mice), the researchers conducted the first gene‑replacement trial in a model of human complex III deficiency.
“We injected adeno‑associated viruses carrying a functional copy of the Bcs1l gene into three‑week‑old mutant mice and showed that this restored complex III function in the liver, preventing liver disease,” explains Rishi Banerjee.
Preventing the loss of liver complex III activity was sufficient to correct systemic metabolic disturbances: the treated mice maintained normal blood glucose levels, grew better, and showed normalized whole‑body metabolism. Survival increased by nearly two‑fold. Notably, liver correction also improved energy metabolic in skeletal muscle, underscoring the central role of the liver in whole‑body energy homeostasis and growth regulation in mitochondrial disorders.
Because most BCS1L mutations in patients cause milder disease than GRACILE syndrome, these findings may have translational relevance to patients. Liver‑directed gene therapy could represent a promising therapeutic strategy for mitochondrial diseases characterized by prominent liver involvement.
Original article:
Hepatic gene replacement improves energy metabolism and extends survival in a mouse model of neonatal mitochondrial disease GRACILE syndrome.
Banerjee R, Purhonen J, Sultana N, Ros O, Nieminen AI, Kietz C, Fellman V, Kallijärvi J. Molecular Therapy 2026.